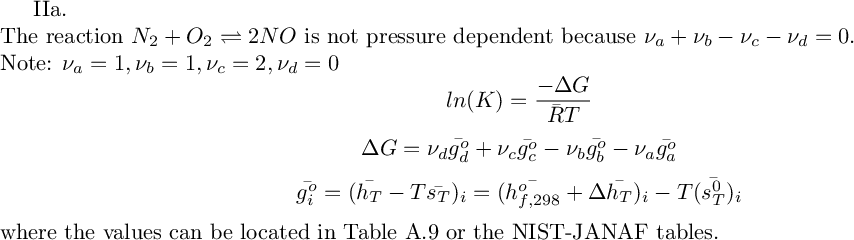
-10.546697881173996
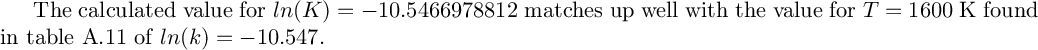
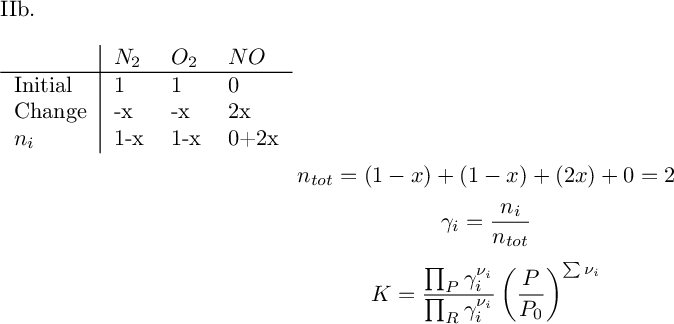
('x=', [0.00255665363707503])
Equilibrium composition is the following:
yN2, yO2, yNO (, , )
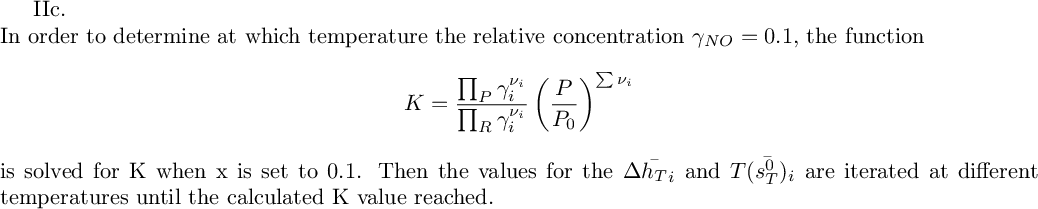
ln(K) when NO is at 10% concentration=
Using Table A.11, find the temperature that corresponds, which is equal to K
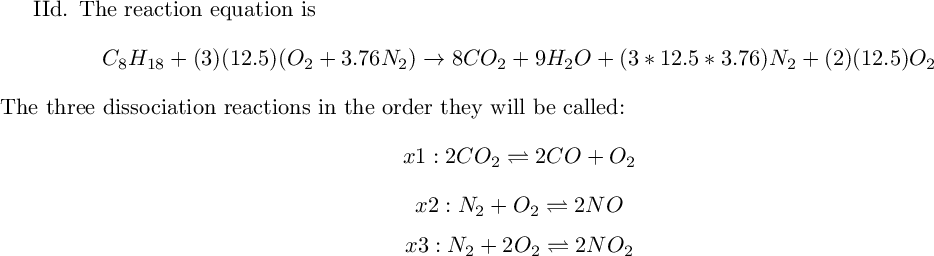
[, , , , , , , , ]
f1
f2
f3
Error in lines 1-2
Traceback (most recent call last):
File "/projects/sage/sage-6.10/local/lib/python2.7/site-packages/smc_sagews/sage_server.py", line 904, in execute
exec compile(block+'\n', '', 'single') in namespace, locals
File "", line 2, in <module>
File "sage/symbolic/expression.pyx", line 4811, in sage.symbolic.expression.Expression.substitute (/projects/sage/sage-6.10/src/build/cythonized/sage/symbolic/expression.cpp:27729)
self._gobj.subs_map(smap, 0))
ValueError: power::eval(): division by zero